MINERAL OIL PROPERTIES
This article discusses gassing properties of transformer insulating oils, correlating the Hα content of a number of gassing inhibitor compounds with the gassing tendency of mineral transformer oils.
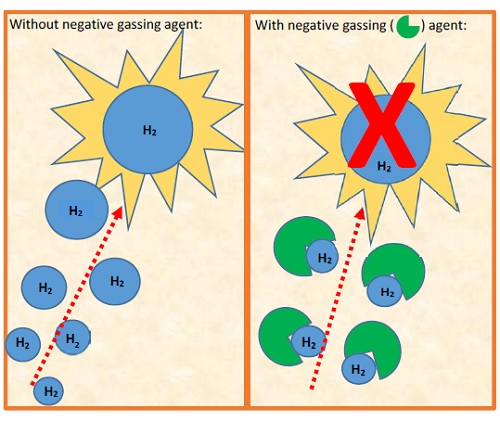
In transformers, oil in combination with paper is used to provide insulation to the transformer’s windings and other internal components. The majority of transformer insulating oils are composed of base oils and specific performance additives. Gassing inhibitors may be added to higher quality base oils to improve the gassing properties of the finished insulating oil. An insulating oil with a negative gassing tendency reduces hydrogen gas bubbles which result from electrical and thermal fault conditions in the transformer.
A negative gassing tendency also reduces the risk of transformer failure and explosion due to hydrogen gas generation, providing an extra margin of safety. As a chemical class, gassing inhibitors are also known as hydrogen donors. Structurally, chemical compounds containing Hα protons, protons attached to non-aromatic carbon atoms that are attached directly to an aromatic ring structure, are potential hydrogen donors. In this study, the Hα content of a number of gassing inhibitor/hydrogen donor compounds were measured using 1H-NMR. The correlation between Hα proton content and the gassing tendency of a transformer insulating oil was then studied using the ASTM D2300 test method.
Introduction
Transformers are critical and expensive pieces of equipment in the power generation and distribution network. Any failure could have drastic consequences to an entire electrical network. To protect and increase the life of transformers, insulating oils are used in combination with solid insulation. Together, they provide dielectric insulation to the transformer’s windings and internal components and, also, dissipate heat generated by the transformer’s windings and core [1, 2].
Different types of insulating liquids exist, including petroleum derived mineral oils, natural and synthetic esters, silicone, and synthetic hydrocarbons.
Mineral oil based transformer/insulating oils are commonly composed of base oil and additives that enhance oxidation stability, cold temperature performance, and gassing properties of the base oil. The base oil may be an isoparaffinic oil or naphthenic oil. Isoparaffinic oils are produced through a severe hydrotreating process; as a result of the process, deleterious sulfur, nitrogen, and oxygen compounds are eliminated, and undesirable aromatic compounds are converted to more stable compounds. This study used isoparaffinic mineral transformer oils for their superior dielectric and oxidative stability, as well as established performance with gassing inhibitors.
Although the hydrocarbons in finished mineral oils are carefully refined and highly stable, the molecules may decompose through thermal or electrical stresses. During a thermal or electrical fault, energy is released and absorbed by the mineral oil’s hydrocarbon molecules. This excess energy breaks down chemical bonds in the hydrocarbons, producing hydrogen gas alongside low molecular weight hydrocarbons [3].
The tendency of an oil to absorb or evolve gas under electrical stress can be measured by ASTM D2300 (standard test method for gassing of electrical insulating liquids under electrical stress and ionization).

It’s these characteristic hydrocarbons that are monitored by routine dissolved gas analysis (DGA) testing to monitor the health and performance of the transformer. The remaining part of the decomposed hydrocarbon is a free radical, which can react with a similar free radical and produce higher molecular weight species which are no longer soluble in the oil (i.e. sludge). When this process happens, the evolved hydrogen gas exacerbates oil aging, and is a potential threat to the operational safety of the transformer.
The tendency of an oil to absorb or evolve gas under electrical stress can be measured by ASTM D2300 (standard test method for gassing of electrical insulating liquids under electrical stress and ionization). In this test method, electric stresses are simulated by ionic bombardment of the oil molecules producing hydrogen and other breakdown species. Should they be present, gassing inhibitors can absorb the generated hydrogen gas in the insulating oil. The value reported by ASTM D2300 is the net value of these two competing reactions. A positive result indicates gas is evolved following electrical stress. If gas is absorbed, however, the result is a negative gassing tendency. During this equilibrium, saturated hydrocarbons are mainly responsible for the hydrogen gas produced. On the other end, aromatic molecules, or unsaturated bonds within a molecule, tend to absorb free hydrogen.
Since isoparaffinic mineral oils are highly refined to enhance their oxidative stability, they tend to evolve hydrogen under stress as they lack reactive unsaturated and aromatic molecules. To improve the gassing tendency of stable isoparaffinic mineral oils without compromising their oxidation stability, gassing inhibitors are added to the oil. As mentioned, gassing inhibitors are also known as hydrogen donors, as they can donate a hydrogen bond with free hydrogen molecules. A hydrogen donor may be any compound which contains labile or easily displaced hydrogen. By readily displacing existing hydrogen bonds, the gassing inhibitor is able to absorb new, free hydrogen.
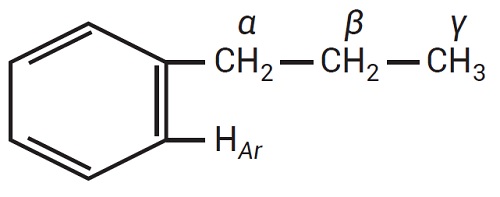
Figure 1. Hydrogen types in a molecule
Examples include partially saturated aromatic compounds such as tetrahydronaphthalene, alkyl substituted tetrahydronaphthalenes, and alkylated benzenes [4]. The ability of a donor material to donate a hydrogen bond can be expressed in terms of specific types of hydrogen content. There are four hydrogen types according to the 1H-NMR (Nuclear Magnetic Resonance) spectral analysis of hydrocarbons, as shown in Table 1. For a better visualization, the four hydrogen types are shown in Figure 1 [5, 6].
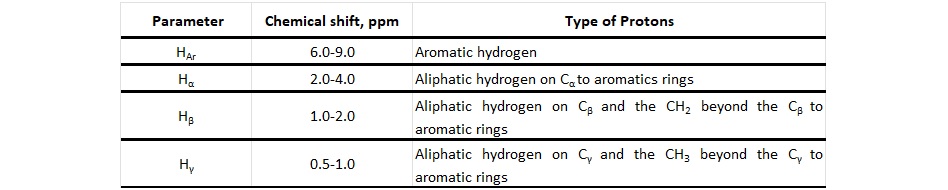
Table 1. Hydrogen types and chemical shift
The HAr protons are attached to aromatic rings and are a measure of the aromaticity of a material. Hα protons are labile and are potential hydrogen donors. They are attached to non-aromatic carbon atoms attached directly to an aromatic ring structure.
Hβ -protons are attached to carbon atoms in a second position away from an aromatic ring. Protons attached to carbon atoms three or more bonds away from an aromatic ring structure are Hγ protons.
As such, the performance of any hydrogen donor was expected to be linked to the type and amount of aromatic structures, alongside higher Hα proton content [5].
Experimental
To study the effectiveness of different hydrogen donor compounds as gassing inhibitors, 1H-NMR was first used to measure HAr and Hα proton content in four different compounds. The gassing tendency of transformer oils made with each compound was then measured using the ASTM D2300 test method to assess the correlation between the two properties.
Four hydrogen donor streams (HD1 to HD4) were assessed in this study of which three were pure compounds and one was a mixed hydrocarbon refinery byproduct. 1H NMR spectra were recorded at room temperature on a Bruker Avance II 400 spectrometer.
Approximately 5-10 mg of sample was dissolved in approximately 1 mL of CDCl3 (Cambridge Isotope Laboratories, 99.8% D, w/ 1% V/V TMS) in a 5 mm NMR tube. 1H (400.32 MHz) experiments were run with an interscan delay of 4.83 s and 32 scans were collected.
Figure 2 shows the 1H-NMR spectra for each hydrogen donor.
Gassing inhibitors, also known as hydrogen donors, are used to improve the gassing tendency of stable isoparaffinic mineral oils without compromising their oxidation stability.
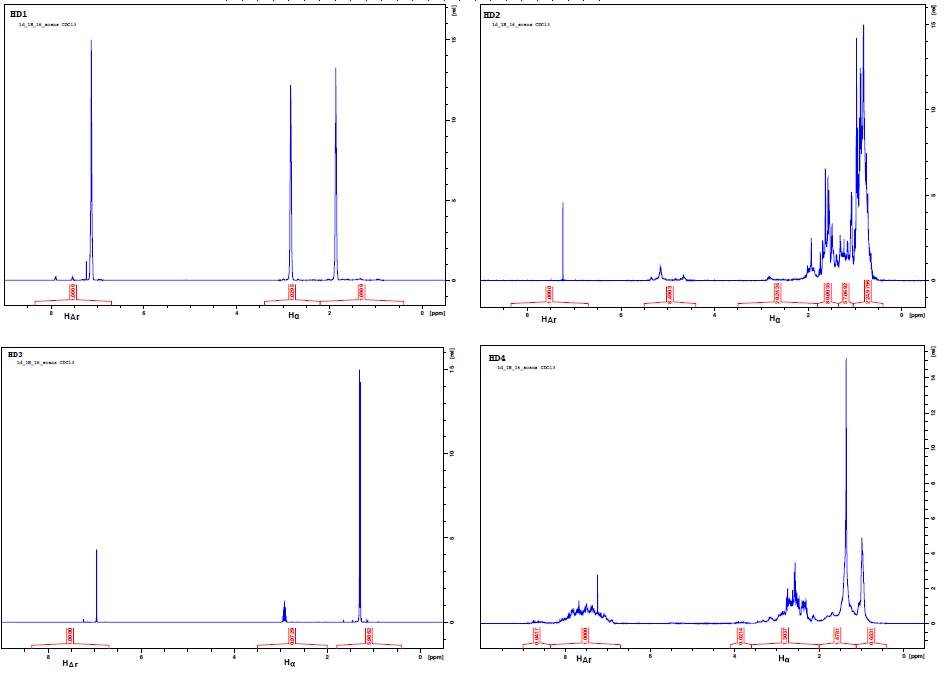
Figure 2. 1H-NMR Spectra
Results and Discussions
For 1H-NMR interpretation, the typical 1H-NMR signal positions reported in Table 1 were used. The percentage of Hα and HAr protons was calculated for the hydrogen donor compounds tested in this study and is shown in Table 2. Three different samples of the HD4 stream were tested to check the variation between different production batches.
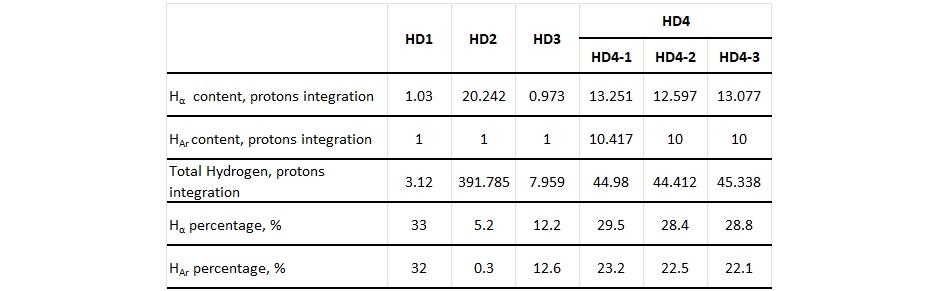
Table 2. Hydrogen content of four different hydrogen donor compounds using 1H-NMR data
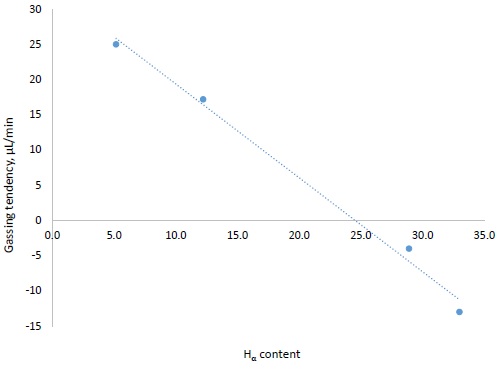
The gassing tendency of transformer oil blends made using each hydrogen donor as the gassing inhibitor were then measured by the ASTM D2300 test method. Table 3 shows the gassing tendency results. The treat rate of the hydrogen donor/gassing inhibitor was the same in all the blends at 2 vol.%. Results show that hydrogen donor material with Hα content greater than 28% provides the blend with a negative gassing tendency. The elevated amount of HAr for the compounds with higher Hα content indicates the link between hydrogen donor performance and aromatic structures, alongside with higher Hα proton content.
Negative gassing tendency is a unique property for the transformer oil because it reduces the risk of transformer failure and explosion due to hydrogen generation, providing an extra margin of safety and performance.
Figure 3 shows the correlation between Hα content of a hydrogen donor and the gassing tendency of an isoparaffinic transformer oil containing 2 vol.% hydrogen donor material.
Figure 3. Gassing tendency of a transformer oil vs Hα content of hydrogen donor compound used in the transformer oil
Results confirm that the addition of a superior hydrogen donor additive, based on Hα content, provides superior negative gassing properties to the oil.
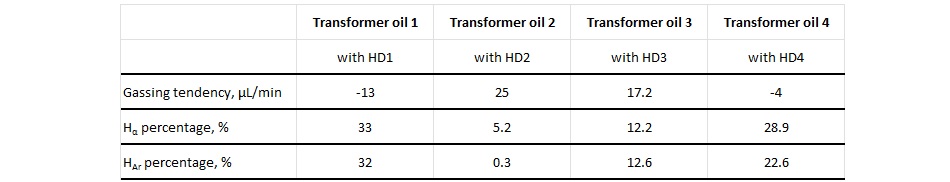
Table 3. Gassing tendency results
Conclusions
The effectiveness of different hydrogen donor compounds as gassing inhibitors in isoparaffinic transformer oils was studied. The 1H-NMR technique was used to assess the Hα and HAr content of different hydrogen donor compounds. The gassing tendency of a representative transformer oil blend containing 2 vol% of each hydrogen donor was then measured using the ASTM D2300 test method.
The correlation between Hα content and gassing tendency indicates higher Hα content provides negative gassing tendency to the transformer oil.
Negative gassing tendency is a unique property for the transformer oil because it reduces the risk of transformer failure and explosion due to hydrogen generation, providing an extra margin of safety and performance.
References
[1] I. Fofana and J. Sabau, “Application of Petroleum-based oil in Power Transformer,” Natural Gas Research Progress, Editors: N. David and T. Michel, Nova Science Publishers, Inc., ISBN: 978-1-60456-700-7, 2008
[2] D. Peterchuck and A. Pahwa, “Sensitivity of transformer’s hottest-spot and equivalent aging to selected parameters,” IEEE Trans. Power Delivery Vol. 17, pp. 996-1001, 2002
[3] J. S. N’Cho, I. Fofana, A. Beroual, T. Aka-Ngnui and J. Sabau, “The Gassing Tendency of Various Insulating Fluids under Electrical Discharge,” IEEE Transactions on Dielectrics and Electrical Insulation, Vol. 18, No. 5, pp.1616-1625, 2011
[4] M. Fefer and T. Ruo, “Dielectric Fluid,” US patent 67, 790, 386 B2, 2004
[5] L. R. Rudnick, “Process for Visbreaking resins in the Presence of Hydrogen-Donor Materials and Organic Sulfur Compounds,” US Patent 4, 587, 007, 1986
[6] C. O. Rossi, P. Caputo, G. De Luca, L. Maiuolo, S. Eskandarsefat and C. Sangiorgi,“ 1H-NMR Spectroscopy: A Possible Approach to Advanced Bitumen Characterization for Industrial and Paving Applications,” Applied Science, 8, 229, 2018